|
Case of the Month: February 2026
Diagnosis and Discussion
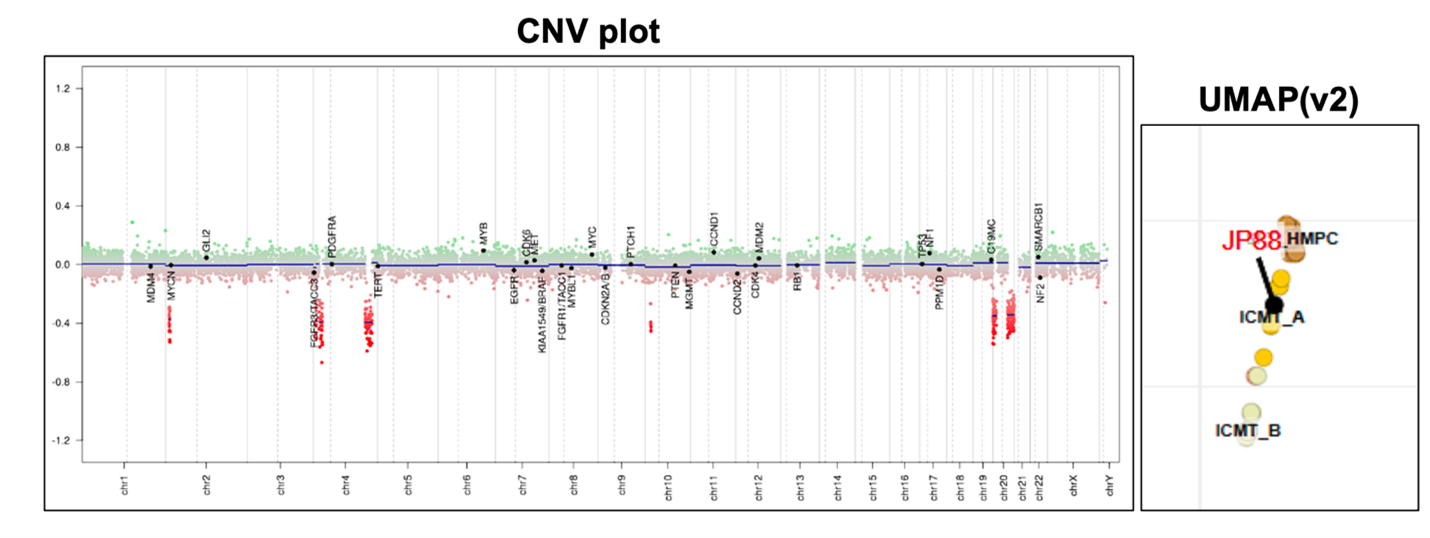
The Final diagnosis for above entity is as below: Integrated diagnosis: Intracranial mesenchymal tumor, FET::CREB fusion positive Histopathological diagnosis: Overall histopathological features favor CNS Mesenchymal tumor CNS WHO grade: Not assigned Molecular Information: EWSR1::ATF1 fusion identified.
Intracranial mesenchymal tumor with FET::CREB fusion is a recently described (a provisional entity), primary intracranial mesenchymal neoplasm in children and young adults characterized by fusion of a FET RNA-binding protein family gene (usually EWSR1, rarely FUS) with a member of the CREB family of transcription factors (CREB1, ATF1, or CREM) with variable histomorphology and have been variously termed intracranial angiomatoid fibrous histiocytoma or intracranial myxoid mesenchymal tumor. The identified fusions on sequencing included EWSR1::ATF1, EWSR1::CREB1, EWSR1::CREM, FUS::CREM and SMARCA2::CREM and are divided into 2 subclasses A (mostly EWSR1-ATF1 and EWSR1-CREB1 fusions) and B (enrichment for tumors with CREM as the fusion partner, either EWSR1-CREM or FUS-CREM). Epidemiology Occurs more frequently in children or young adults with a predominance in the female population, although cases in adults in their fifties and sixties have been reported. clinical presentation Symptoms of headache, nausea, vomiting, tinnitus, and occasionally seizures or focal neurological deficits due to mass effect and specific location. Anemia or hemorrhage have been reported. Anatomical localization: Tumors are uniformly extra‐axial or intraventricular and localizations at the cerebral convexities, falx, lateral ventricles, tentorium, cerebellopontine angle, and spinal cord have been reported. They are more commonly located in supratentorial sites than in infratentorial sites. Group A tumors most often occurred in adolescence or early adulthood, arose throughout the neuroaxis, and contained mostly EWSR1-ATF1 and EWSR1-CREB1 fusions. Group B tumors arose most often in early childhood, were located along the cerebral convexities or spinal cord, and demonstrated an enrichment for tumors with CREM as the fusion partner (either EWSR1-CREM or FUS-CREM). Radiology IMT FET::CREB fusion tumors are usually circumscribed, extra-axial tumors with dural attachment and compression of the subjacent brain parenchyma. Lobulated growth (often with both solid and cystic components), avid enhancement after contrast administration with solid and cystic growth, intratumoral blood products, and substantial peritumoral oedema are other radiological findings. Can mimic meningioma by demonstrating a dural tail or bony involvement of the overlying skull. Etiology and Pathogenesis: No known association with familial tumor predisposition syndromes. The etiology and cell of origin is unknown. These tumors harbor fusion of a FET RNA-binding protein family gene (mostly EWSR1) with a member of the CREB family of transcription factors: CREB1, ATF1 or CREM. Identical FET‐CREB fusions are recurrently found in clear cell sarcoma of soft tissue, extracranial angiomatoid fibrous histiocytoma, gastrointestinal clear cell sarcoma, primary pulmonary myxoid sarcoma, hyalinizing clear cell carcinoma of the salivary gland and a subset of malignant mesotheliomas lacking BAP1 and NF2 alterations, However, the exact relation of these FET‐CREB fusion‐driven neoplasms remains unknown. SMARCA2::CREM fusion has also been reported in few cases. Gross findings: Well-circumscribed, solid, and cystic masses, partially encapsulated, focally hemorrhagic, tan-brown, and focally gelatinous. Histologic features: Variable morphological features including spindle cells, mucin-rich stroma, haemangioma-like vasculature, or epithelioid cells in a mucin-poor collagenous stroma have been described. Dense lymphoplasmacytic cuffing at the tumor periphery or along fibrous septa and haemosiderin or haematoidin are often present with additional morphological features can include meningothelial-like whorls and amianthoid-type fibres. Architecture ranges from syncytial or sheet-like growth to reticular cord-like structures, and fibrous septa separating nodules of tumor cells in subset of tumors. Demonstration of a diagnostic FET::CREB family gene fusion FISH or DNA/RNA sequencing strategies is required for the definitive diagnosis. Tumors with EWSR1::CREB1 fusions more often have stellate/spindle cell morphology, mucin-rich stroma, and haemangioma-like vasculature, whereas tumors with EWSR1::ATF1 fusions are more commonly composed of sheets of epithelioid cells with mucin-poor collagenous stroma. Tumor cells are consistently positive for desmin (sometimes focally), CD99 and EMA and are negative for skeletal muscle markers (myogenin and MyoD1), smooth muscle markers (SMA, caldesmon, and calponin), melanocytic markers (S100, HMB45), glial markers (GFAP and OLIG2), CD34 and STAT6. CD68 and CD163 have been positive in both neoplastic cells and tumor‐associated macrophages. SMARCB1 (INI1) and SMARCA4 (BRG1) expression is retained. Ki67 Proliferation index is generally low. Collagenous stroma with dense intercellular matrix highlighted by reticulin staining. Molecular Findings: Demonstration of a diagnostic FET::CREB family gene fusion FISH or DNA/RNA sequencing strategies is required for the definitive diagnosis and may be detected using FISH or DNA/RNA sequencing strategies. Demonstrating EWSR1 rearrangement via break-apart FISH assay is not specific in isolation, and confirmation of a CREB family fusion partner is recommended. Significant CREM over expression regardless of fusion type has been reported, when compared to either meningioma alone, or a larger group of CNS tumors in gene expression analysis of ICMT. Differential diagnosis: The differential diagnosis is usually with extracranial or metastatic sarcomas or atypical meningioma especially of chordoid, rhabdoid and microcystic types. Other differential diagnosis are Solitary fibrous tumor and other CNS Mesenchymal tumors. Pathognomonic gene fusion requires for the diagnostic confidence. Prognosis: Intracranial mesenchymal tumor with FET::CREB fusion neoplasm is prone to tumor recurrence and there are two prognostically relevant methylation subclasses. It has been reported that patients with methylation subclass B tumors have shorter progression‐free survival relative to subclass A tumors (median 4.5 vs. 49 months, p = 0.001). The full spectrum of clinical behavior of ICMT is not yet known, but it ranges from slow growth to rapid recurrences and rarely, cerebrospinal fluid dissemination or systemic metastases, including pulmonary and thoracic lymph nodes and bony metastases to spine have been reported. The diagnostic distinction of this tumor from other epigenetically and histologically related tumors are required. References: 1. Intracranial mesenchymal tumour, FET::CREB fusion-positive. The 2021 Who classification of the central nervous system (5th ed.). IARC Press. Lyon, Fr. 2021 2. Sloan EA et al. Intracranial mesenchymal tumor with FET‐CREB fusion—A unifying diagnosis for the spectrum of intracranial myxoid mesenchymal tumors and angiomatoid fibrous histiocytoma‐like neoplasms. Brain Pathol. 2021 Jan 28;31(4):e12918. doi: 10.1111/bpa.12918 3. Rajan S. et al. Intracranial mesenchymal tumor, FET::CREB fusion-positive: an integrative analysis of 81 cases. Neuro Oncol. 2026 Jan 22:noag001.doi: 10.1093/neuonc/noag001. 4. Komatsu M. et. al. Intracranial myxoid mesenchymal tumor with EWSR1-CREB1 gene fusion: a case report and literature review. Brain Tumor Pathol. 2020 Apr;37(2):76-80. 5. Konstantinidis A et al. Intracranial Angiomatoid Fibrous Histiocytoma with EWSR1-CREB Family Fusions: A Report of 2 Pediatric Cases. World Neurosurg. 2019 Jun:126:113-119. doi: 10.1016/j.wneu.2019.02.107.Epub 2019 Mar 2. 6. Bale TA et al.Intracranial myxoid mesenchymal tumors with EWSR1-CREB family gene fusions: myxoid variant of angiomatoid fibrous histiocytoma or novel entity? Brain Pathol. 2018 Mar;28(2):183-191.doi: 10.1111/bpa.12504.Epub 2017 Apr 11. 7. Kao YC et al.EWSR1 Fusions With CREB Family Transcription Factors Define a Novel Myxoid Mesenchymal Tumor With Predilection for Intracranial Location. Am J Surg Pathol. 2017 Apr;41(4):482-490. doi: 10.1097/PAS.0000000000000788. 8. Hansen JM et al. Primary intracranial angiomatoid fibrous histiocytoma presenting with anaemia and migraine-like headaches and aura as early clinical features. Cephalalgia. 2015 Dec;35(14):1334-6.doi: 10.1177/0333102415583988.Epub 2015 Apr 21. 9. Sciot R et al. Primary myxoid mesenchymal tumour with intracranial location: report of a case with a EWSR1-ATF1 fusion. Histopathology. 2018 Apr;72(5):880-883.doi: 10.1111/his.13437.Epub 2018 Jan 11. |